Mono Ammonium Phosphate
Grades We Manufacture :
- Mono Ammonium Phosphate (Industrial Grade)
- Mono Ammonium Phosphate (LR/Pure Grade)
Formulation :
H3PO4 + NH3 = (NH4)H2PO4
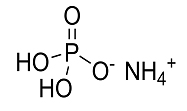
Mono ammonium phosphate is formed when ammonia gas is added tosolution of phosphoric acid and reacted until the desired PH is obtained and solution gets distinctly acidic. End product is in the form of tetragonal prisms.
Application :
- Mono ammonium phosphate is used as a baking powder with sodium bicarbonate
- Widely used in fireproofing of paper, wood, fiberboard
- Mono ammonium phosphate is mixed with ammonium sulfate and resultant is used in ABC dry chemical fire extinguishers
- MAP is also used in fermentation of yeast
- MAP provides a rich source of phosphate and nitrogen usable by plants helping their growth. It contains relatively less PH than di ammonium phosphate which suits it best for fertilizer use, reducing the seed damage
Specifications :
| Product Name | Mono Ammonium Phosphate (MAP) |
| IUPAC Name | Ammonium Di-hydrogen Phosphate |
| Molecular Formula | (NH4)H2PO4 |
| Molecular Weight | 115 |
| Appearance | White crystals |
| PH Range (5 % w/v) | 4.0 – 5.0 |
| Assay % / Purity % (minimum) | 97.00 |
| P2O5 Content % (minimum) | 61.00 |
| ‘N’ Content % (minimum) | 12.00 |
| Iron AS ‘Fe’ content % | 0.02 |
| Chloride as ‘Cl’ content % | 0.2 |
| Sulfate as ‘SO4’ content % | 0.03 |
Note: All of our products are manufactured in
(A)Technical Grade for industrial use, water treatment, enzyme process, several animal feed, buffering agents in several pharmaceutical process, higher agriculture grade fertilizers, and other uses, as well as we manufacture
(B) Pure grade which successfully qualify to all pharmaceutical, AR, Indian Pharmacopoeia, British Pharmacopoeia, United States Pharmacopoeia, Food, Animal Feed, Seafood and laboratory reagent grade standards. This is to meet all the standards of quality in any grade required by customer.
Packing :
- HDPE bag with LDPE liner in 20 kg, 25kg, 40kg and 50 kg
- As per customer's request


